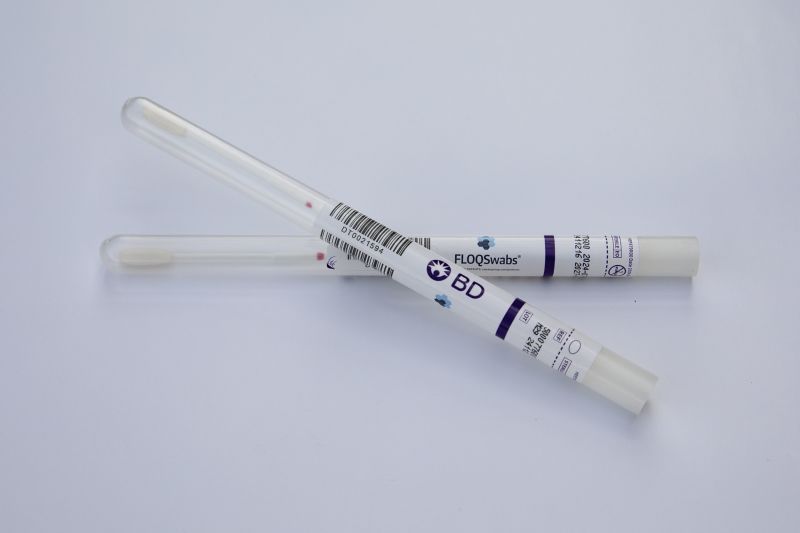
The first shipments of some self-collection HPV tests for cervical cancer screening are currently on their way to doctors’ offices across the United States.
In May, the US Food and Drug Administration gave the greenlight for patients to have the option to collect their own vaginal samples for cervical cancer screenings instead of undergoing traditional HPV tests or Pap smears, which involve a speculum to screen for the disease. Similar to how they may collect their own urine samples, patients can now collect their own vaginal samples in a health-care setting, such as a doctor’s office, urgent care or pharmacy clinic.
So far, two health care businesses – biotechnology company Roche and medical technology firm BD – already have HPV tests that can be used with self-collected samples. Most cervical cancer screenings involve testing for human papillomavirus or HPV, as most cervical cancers are caused by the virus.
Shipments of the BD self-collection HPV tests began Thursday, according to the company. And Roche anticipates that its HPV self-collection screening solution will be shipped sometime this fall.
The hope is that providing a self-collection method will make screening more accessible for patients, leading to more women being screened, said Dr. Jeff Andrews, board-certified gynecologist and vice president of Global Medical Affairs for Diagnostic Solutions at BD.
Each year in the United States, more than 11,000 new cases of cervical cancer are diagnosed and about 4,000 women die of the disease, according to the US Centers for Disease Control and Prevention. It’s estimated that about half of invasive cervical cancer cases are diagnosed in people who have never been screened and about 10% of diagnoses are in people who had not been screened in the five years prior.
“In other countries in Europe, Australia, New Zealand, we know that adding the option of self-collection has reached some of those women who were previously under screened. So that’s the main goal, is to develop a methodology that would appeal to persons with a cervix who would otherwise not be screened,” Andrews said.
“In the United States in particular, almost 30% of women are unscreened or under screened,” he said. “Those women are the ones most likely to have cervical cancer. About two-thirds of cervical cancers occur in women who are not screened within the interval,” as in the recommended time interval for which women are recommended to screen.
Methods to screen for cervical cancer
The US Preventive Services Task Force recommends screening for cervical cancer with cervical cytology– also known as a Pap test or Pap smear –every three years for women ages 21 to 29. For women ages 30 to 65, the USPSTF recommends screening every three years with cervical cytology alone, every five years with high-risk HPV testing alone, or every five years with high-risk HPV testing in combination with cytology.
Typically, gynecologists collect samples for HPV testing, a cervical cytology or both – and any of those screening methods, in which samples are collected, can feel uncomfortable for some patients when they involve a speculum.
Cervical cytology involves examining cervical cells for changes to find precancerous or cancerous cells. The HPV test checks cells for infection with the high-risk types of HPV that can cause cervical cancer. An HPV and Pap co-test uses both tests together to check for both high-risk HPV and cervical cell changes.
It’s estimated that about 80% of people will get an HPV infection in their lifetime. HPV, a group of more than 150 viruses, is spread primarily through sexual contact and includes low-risk strains, which most often cause warts, and high-risk strains, which have been associated with an increased risk of certain cancers, such as cervical, anal, penile and oropharyngeal cancers. In most cases, HPV clears on its own within two years, but when the infection does not go away, health problems like cancer may occur.
“Almost all cervical cancers are caused by persistent infection with certain types of HPV,” Dr. Karen E. Knudsen, CEO of the American Cancer Society, said in a news release in May when the FDA approved HPV self-collection testing for cervical cancer screening. “Self-collection can expand access to screening and reduces barriers, which will give more people the opportunity to detect, treat, and ultimately survive cancer.”
For some patients, having a provider collect a vaginal sample for cervical cancer screening or HPV testing can be painful or awkward. The screening process involves a patient spreading their legs, with their feet held in stirrups, as their doctor vaginally inserts a cold, metal speculum so that they can then scrape cells from their cervix.
Many patients may skip recommended Pap exams to screen for cervical cancer because the process of a traditional Pap smear screening can be painful.
Research suggests that having the perception that a Pap test will be painful significantly reduces the likelihood of women setting the goal to schedule their first-ever Pap exam. But minimizing pain during a Pap test will likely increase acceptability of and adherence to cervical cancer screenings.
“If we can increase screening, we can reduce the incidence of this cancer. Having another modality or choice to make screening more accessible and available is important especially for our underinsured and underserved populations. Self-screening offers this extra choice for women,” Dr. John Vullo, chairman of the department of obstetrics and gynecology at Catholic Health’s Good Samaritan University Hospital on Long Island and associate chair of Catholic Health Women’s Health Services, said in an email.
“Although I believe this new choice is important to offer our patients, I am glad the current recommendations are for it to be used under the supervision of a provider so that the patient can truly get and understand the benefits and limitations of this new testing,” Vullo said in part. “This will allow us to offer them another modality to screen for cervical cancer while still giving them the benefit of speaking with a provider to discuss other health issues that are part of the annual well woman exam.”
For the self-collection screening test, a clinician would have to order the test for a patient, and “that is to be sure that the person doing the test will get the results explained to them, so the person now would be in contact with their provider,” Andrews said.
That order would be directed to a lab. The lab would then create a self-collection kit, which would include instructions, a six-inch swab and the screening test itself. The kit would be shipped to the doctor’s office for the patient. The patient would use the swab to self-collect a vaginal sample, by inserting it three inches into the vagina – this could be done in the bathroom at the doctor’s office – and then the doctor’s office would ship the sample to the lab for processing. If a sample tests positive, the doctor would likely recommend further testing.
Home tests are possible in the future
While the self-collection method is now available in health-care settings, companies are hoping the self-collection kits could eventually be cleared for use at home, too.
BD has been working closely with the FDA to provide data supporting the use of its self-collection tests within the home, Andrews said.
- Sign up here to get The Results Are In with Dr. Sanjay Gupta every Tuesday from the CNN Health team.
Another company, Teal Health, has developed an at-home cervical cancer screening device called the Teal Wand that was granted “breakthrough device” status in May by the FDA, which would allow the agency to review the device on a faster timeline.
“FDA’s recognition of the Teal Wand as a Breakthrough device acknowledges the important public health benefit that self-collection for cervical cancer screening can have on those who are rarely screened or who do not participate in clinician-based screening for cervical cancer,” Trena Depel, vice president of clinical and regulatory at Teal Health, said in a news release at the time.
Regardless of where it takes place, screening for cervical cancer remains important because early cases often may not have signs or symptoms. Advanced cases may cause abnormal vaginal bleeding or unusual discharge. Cervical cancer is treated in many ways, including surgery, chemotherapy and radiation therapy.
Some of the most important steps women can take to help prevent cervical cancer, according to the CDC, are to get vaccinated against HPV, not smoke, use condoms during sex, have regular screening tests and go back to see the doctor if screening test results are not normal.